STT. New Lesion Found in Will’s Pelvis as Doctors Evaluate Next Steps in Treatment
WILL’S HEALTH CONDITION IS STABLE, MEPACT IMMUNOTHERAPY IS OFFICIALLY AVAILABLE IN THE US AFTER A 3-MONTH WAITING PERIOD.
After days of waiting for scan results and expert consultations, Will’s family has officially released the latest update on his bone cancer treatment.
According to information from the oncology team, the combined results of scans taken over the past two days show that Will’s condition is generally stable, especially compared to the significant progression recorded last December.
While not a major breakthrough, stability in the context of malignancies is considered a positive sign.
Doctors said that some previously damaged areas even showed signs of reduced activity.
Notably, the suspected areas in the liver now show more calcification and less activity compared to previous scans.
The lung and chest area remained relatively stable, with no significant deterioration observed.

The jawbone area still shows signs of activity on the PET scan; however, this is not a new finding.
Previous scans have also revealed a similar condition, and so far there have been no signs of physical bone damage, which is considered an important factor in monitoring osteosarcoma.
However, a new finding has been discovered in the left pelvic bone.
This area is currently being referred to the diagnostic imaging department for further evaluation.
Doctors will consider the possibility of using targeted radiation therapy if necessary.
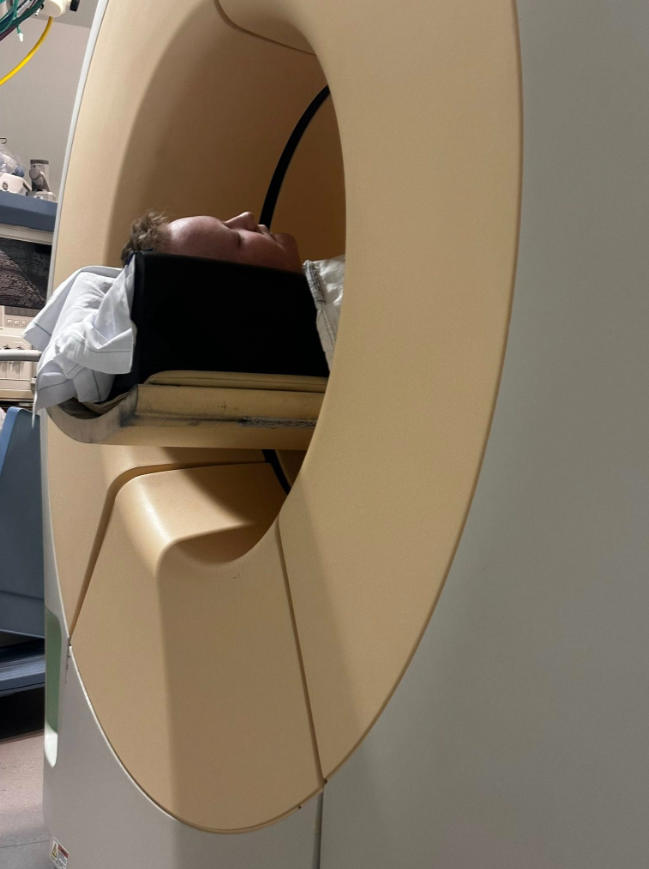
Simultaneously, Will was scheduled for an MRI scan of his spine and pelvis to gain a more comprehensive understanding of his current condition.
The MRI results are expected to be available within 24 hours to support decisions about further treatment.
Meanwhile, a piece of information that the family and medical team considered a significant breakthrough has been officially confirmed.
The immunotherapy drug MEPACT, which had been pre-approved by the U.S. Food and Drug Administration (FDA) for Will due to his severe medical condition, has finally become available in the United States.
Previously, the drug was delayed for three months due to supply shortages from Europe.
MEPACT is currently undergoing the final approval process with the FDA before officially being put into use.
Once approved, Will will begin a twice-weekly treatment schedule at the Children’s Hospital.
The family expressed their gratitude to the medical team for their tireless efforts in seeking and promoting the most advanced treatment options for their son.
In addition, the current treatment plan continues with the Cabo medication for another six weeks.
After this phase, Will will be re-evaluated with imaging to determine the effectiveness and next steps.
Another piece of positive news concerns the arm pain Will has been experiencing recently.
The PET scan results showed no signs of bone cancer spreading in this area.

Doctors identified inflammation as the primary cause and developed a new pain management protocol to improve the child’s quality of life.
The family shared that the days spent scanning for PET were always the most stressful.
However, they are learning to process each piece of information calmly and trust the treatment plan.
They affirmed that their goal remains unchanged: to keep moving forward, to keep fighting, and to maintain their faith.
After a long day at the hospital, the family brought Grandma home and spent the weekend relaxing together.
Although the treatment journey still presents many challenges, Will’s current stability and the preparation for the deployment of a new immunotherapy are seen as important steps in his battle.
The family also extends their gratitude to the community for their prayers, messages, and unwavering support throughout this difficult time.